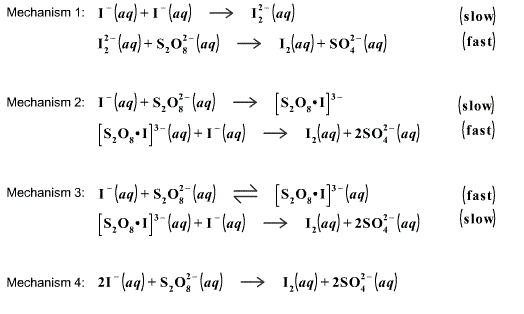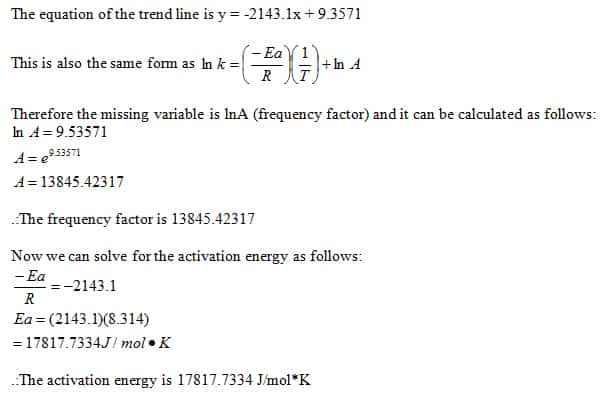What is the reaction order with respect to [S2O8 2-] and [I-] in a persulfate-iodide reaction? - Quora

Autocatalysis-Driven Clock Reaction III: Clarifying the Kinetics and Mechanism of the Thiourea Dioxide–Iodate Reaction in an Acidic Medium | The Journal of Physical Chemistry A

SOLVED: Reaction Rates Concentrations Effect of Reactant Concentrations Demo: "lodine Clock" Reaction lodateVariation I03 + 3 HSO3 > 1-+ 3 S042-+3 Ht (slow) I03 +51 + 6 Ht> 3 12 + 3